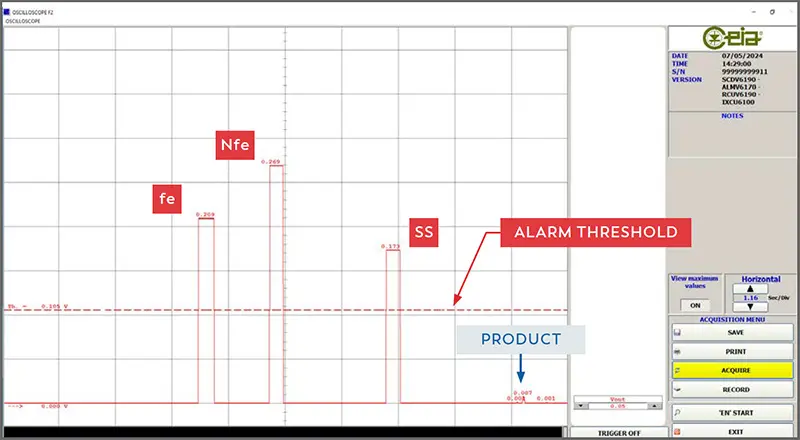
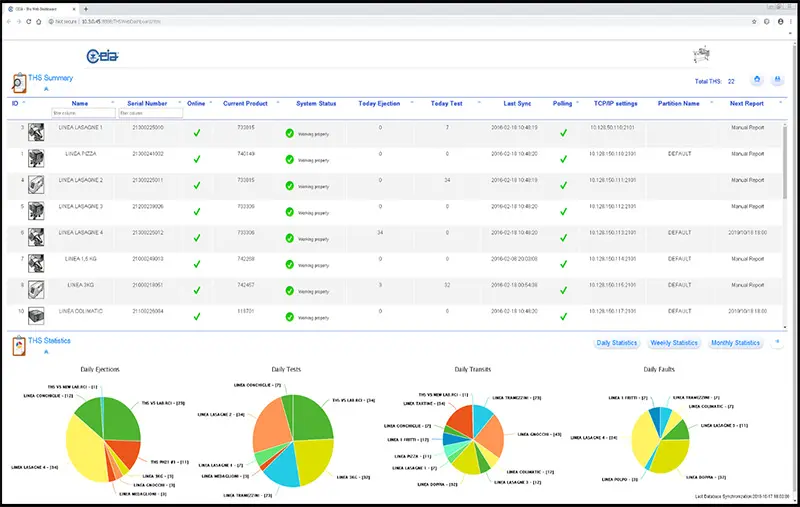
Software diagnostic tool. Allows remote programming, signal analysis and oscilloscope functionalities.
THS/PH21N
Ultra high sensitivity metal detector
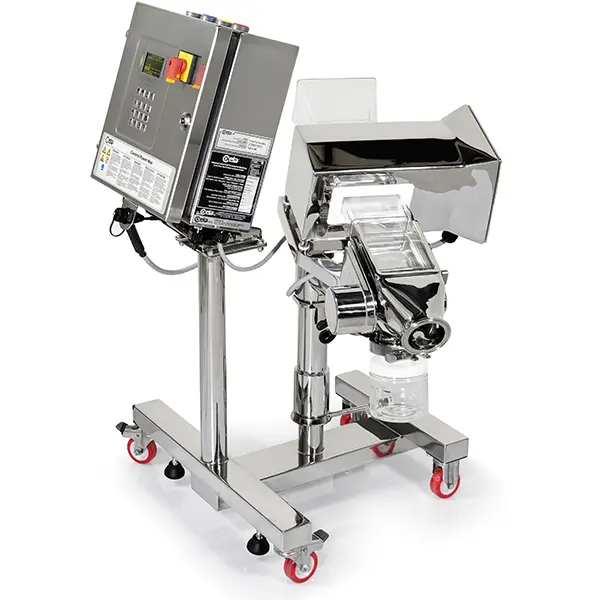
The THS/PH21N is an ultra-high sensitivity Pharmaceutical Metal Detector crafted from AISI 316L stainless steel, specifically engineered to detect magnetic, non-magnetic, and stainless steel contaminants. It ensures superior hygiene and durability, with the capacity to manage 500 product profiles and log up to 100.000 events via the integrated IXC card. The high-contrast OLED display and intuitive interface with "Fast Access" mode streamline daily operations and setup. For maximum operational safety, the system includes built-in AUTOTEST and AUTO-QC functions, with optional UL/CSA compliance. All product-contact components are designed for tool-free disassembly, ensuring rapid maintenance and peak production efficiency.Key Points
Detection Performance and Digital Technology
The THS/PH21N represents the benchmark for ultra-high sensitivity inspection across all magnetic, non-magnetic, and stainless steel metals. CEIA THS systems stand out for their fully digital signal analysis, which guarantees absolute detection independence from electromechanical vibrations and extreme repeatability of responses. Furthermore, the device can be set to 4 different transmission channels (TX) to prevent mutual interference between adjacent units.
Specialized Configurations: DT (Dust Tight) and WIP (Wash In Place)
To meet diverse containment needs, the system offers dedicated solutions. The DT (Dust Tight) configuration features a fully sealed, dust-tight transit tunnel and ejection system, specifically designed to prevent product dispersion into the surrounding environment. For lines requiring frequent washing, the WIP version integrates water injection nozzles to remove post-production residues; in this case, the tunnel and discharge are sealed with OEB 5 certification, ensuring maximum protection.
Construction and Regulatory Compliance
The system complies with 21 CFR Parts 210 and 211 regulations, featuring a mirror finish on all metal parts. Components in contact with the product, made of AISI 316L stainless steel and PETG, can be quickly disassembled without the use of tools. Compliance with FDA 21 CFR Part 11 ensures secure management of electronic records and digital signatures, supported by an SPM Flash memory and a 10,000-event log.
Operational Safety and Ejection Management
Safety is entrusted to Fail Safe mode and a double-confirmation system that monitors the opening and closing of the EO/EC ejection flaps, whose actuation force is programmable. In addition to AUTOTEST and AUTO-QC functions, the Control Power Box is certified according to UL 508A and CSA-C22.2 No. 14-05 standards, with a universal power supply range of 90-253 VAC.
User Interface and Connectivity
The control panel features a stainless steel keyboard and an ultra-high contrast OLED display (3000:1). The system allows the configuration of up to 40 operators with programmable access levels and can store 500 product profiles. Connectivity includes direct RS-232, integrated Bluetooth, and options for Ethernet or Wi-Fi interfaces, facilitating integration into corporate management systems.